Coronavirus Could Bring New Financial Pressures to Distressed Healthcare Industry
3 Pressure Points the Outbreak Might Bring to U.S. Health System
On March 11, the World Health Organization (WHO) declared the coronavirus outbreak a pandemic, with numerous countries—including China, the Czech Republic, Hong Kong, Italy, Slovakia and the U.S.—announcing travel restrictions and public distancing measures.
For healthcare providers in the United States—already facing financial distress—the anticipated domestic escalation of the coronavirus outbreak could have direct and lasting impacts on the health system in the form of supply chain disruptions, leading to drug and medical supply shortages, as well as long-term operational and financial challenges. For example, more than three-fourths (76%) of U.S. providers already have 60 days or less cash on hand, and nearly a quarter (23%) are concerned about government reimbursement risks, according to BDO’s Treating Healthcare Distress, a survey of 100 CFOs at mid-market healthcare providers. Variables like instability around the Affordable Care Act and uncertainty around incoming payments because of fluctuating deductibles and patient copays, meanwhile, will persist.
These are the top three pressure points that the coronavirus could create domestically:
1. Medical device and drug shortages:
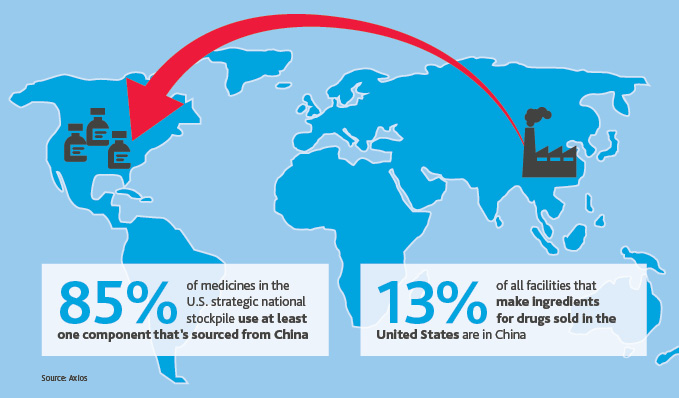
As of Feb. 25, the FDA identified 20 drug products that solely rely on China for their active ingredients or to manufacture their finished drug products. China ranks second among countries that export drugs and biologics to the United States and first for medical devices, according to agency data. China’s position as the world’s largest producer of rare earth minerals—which include elements key to making batteries—adds additional risk, as further economic shutdowns could hinder production of battery-powered medical devices.
Additionally, the United States faces a shortage of medical masks because China is the world’s largest producer of medical masks, though some health officials question their effectiveness.
2. Patient safety risks:
In light of safety concerns and travel limitations, it’s become more difficult for the United States to conduct its regular inspections of drug and medical supply manufacturing sites in China. This could further exacerbate product shortages and patient safety risks associated with less effective products as it limits the U.S.’ ability to oversee quality of production. Additionally, Amazon said on Feb. 20 it had begun removing listings from its online marketplace by sellers claiming to have products that could prevent or kill the coronavirus on the grounds that they were making “medical marketing claims.”
3. Supply chain disruptions:
Outside drug and medical device product shortages, the industry faces other supply chain risks because of its growing practice of offshoring certain operations overseas—notably to companies located in India, China and Vietnam, near or at the epicenter of the outbreak. Billing functions are the ones most frequently offshored in the name of cost and efficiency. (Increasing labor costs is cited as the primary workforce challenge by more than a quarter of healthcare CFOs, according to the 2020 BDO Middle Market CFO Outlook Survey.)
But healthcare organizations in recent years have also begun offshoring other functions like HR and certain electronically enabled diagnostic consultations that don’t require clinicians with U.S. credentials. In the case when an organization’s international vendor has halted operations due to travel restrictions and required at-home employee quarantines, that organization may not be able to execute on certain billing, HR or clinical procedures which could have negative repercussions both financially and operationally.
How U.S. Healthcare Organizations Can Secure Their Operations to Better Support Patient Care
Uncertainty still abounds on whether and how the virus could mutate as well as how many waves of the outbreak might take place. And with the Centers for Disease Control and Prevention (CDC) warning that it’s no longer a matter of if but when the coronavirus spreads further within the United States, patient care is priority #1. Still, for U.S. healthcare organizations, business continuity management is critical, and they must have a proper enterprise risk plan in place sooner rather than later to ensure they can provide effective services to patients.
Organizations should consider the following steps:
- Conduct a proactive business continuity risk assessment: Organizations should identify potential internal operational, financial and market risks; determine direct and indirect impacts; and generate an action plan. It’s critical that action plans incorporate the CDC’s new guidance for how healthcare workers should test for coronavirus. Vulnerabilities via partners on the care continuum or third-party vendors should also be incorporated into action plans.
- Identify a response team to lead ongoing crisis management efforts, coordinating with appropriate federal, state and local authorities, respectively: These efforts should include regular communication to internal and external stakeholders—with patients prioritized. In the event of an emergency, communication with patients, employees (particularly clinicians responsible for containing the outbreak) and partners on the care continuum is key. When it comes to patient communication, organizations should educate not only their direct patients but also their surrounding communities on what coronavirus is and key protective measures people can employ. Leveraging information from the WHO’s dedicated public advice page is a good place to start.
- Regularly monitor announcements from the FDA around supply chain risks and recommended courses of action: The FDA has been releasing regular statements on how the agency is working to diagnose, treat and prevent the disease, as well as monitoring the medical supply chain for shortages, disruptions or fraud risk. Organizations should keep up to date with agency announcements and be ready to adjust their enterprise risk plan in tandem.
- Collaborate with and proactively diversify the locations of your suppliers and vendors: Work closely with your drug and medical supply providers to build scenario models to determine ways to mitigate any additional risks to your supply chain. Identify ways to diversify your supply chain if possible and assess the cost-benefit of maintaining duplicative providers versus the costs of a lost supply chain. In the case of third-party billing or other vendors impacted, organizations would need to have a backup plan in place should vendors be unable to deliver on services.
- Review your organization’s insurance policy: It’s crucial that healthcare organizations understand how to determine and capture lost revenue and income as a result of this unpredictable and unforeseen outbreak. Reviewing coverage around infectious disease, trade disruption and workforce loss is critical to proactively managing the short- and long-term risks.
- Review policies and procedures around the HIPAA Privacy Rule: On Feb. 5, the Department of Health and Human Services (HHS) reminded HIPAA-covered entities of the ways they can share patient information during infectious disease outbreaks. Organizations should ensure they—and their partners on the care continuum—have the proper information governance, cybersecurity and data privacy controls in place to mitigate increased risk around greater sharing of patient information. (This is especially critical to an industry where at least one in five organizations say a data privacy breach is their biggest threat, according to BDO data.)
- Ensure integration of the latest medical billing codes around coronavirus detection: On Feb. 13, the Centers for Medicare & Medicaid Services announced new billing codes for providers and labs to test patients for coronavirus. The codes help ensure organizations can specifically track the public health response to the outbreak—and help mitigate incorrect patient healthcare costs. Organizations should ensure their billing processes have been updated to account for these new codes and that billing staff—domestic and overseas—understand how to correctly bill against them.
- Provide refreshed training to clinicians on telehealth services and best practices: Ahead of anticipated quarantines or inability of certain patients to travel to facilities for in-person medical visits, organizations should review their telehealth capabilities and ensure all clinicians are familiar with the services at their disposal. Leveraging such services could be critical to addressing access to care issues that the outbreak could exacerbate.
- Maintain contemporaneous documentation: While patient safety should be the priority, organizations must ensure they have a designated team—and top down trainings—in place to keep careful records during operational disruptions. Email records around market conditions, cancellations of supply shipments or suppliers being impacted are crucial to preserve as they can be critical to a business interruption claim.
SHARE